“Look a-like, sound a-like” (LASA) refers to a significant issue within the pharmaceutical industry where medications bear similar names or appearances but differ in their active ingredients, dosage forms, strengths, or indications. Look-alike medicines appear visually the same with respect to packaging, shape, colour and/ or size, while sound-alike medicines are similar in the phonetics of their names, doses and/or strengths. These similarities can lead to confusion among healthcare providers, pharmacists, and patients, potentially resulting in medication errors that compromise patient’s health.
LASA incidents highlight the critical importance of clear labelling, distinctive packaging, and effective communication strategies to prevent misunderstandings and ensure the correct administration of medications. LASA errors can occur at any stage of medication use: prescribing, transcribing or documenting, dispensing, administering and monitoring.
LASA occurrences are a global concern, prompting regulatory agencies to implement stringent guidelines to mitigate risks. These measures include requirements for unique drug names, standardized labelling formats, and rigorous pharmacovigilance practices to monitor and address potential issues promptly. Healthcare professionals play a crucial role in identifying and reporting LASA incidents to enhance patient safety and improve medication management practices.
This newsletter delves into the multifaceted issue of “Look-alike, Sound-alike” (LASA) drugs, exploring how similarities in drug names and appearances can lead to medication errors. We examine the impact of LASA drugs on patient safety, regulatory challenges, and the role of intellectual property in the pharmaceutical industry. The article provides an overview of recent regulatory efforts in India, judicial decisions to tackle the issues with LASA drugs, and the need for comprehensive solutions, including technological interventions and enhanced pharmacovigilance. Through this analysis, we aim to highlight ongoing challenges and propose actionable strategies to mitigate the risks associated with LASA drugs.
LASA Drug Issues and Trademark Law
In the pharmaceutical sector, Intellectual Property (IP) rights play a crucial role in incentivizing innovation and protecting investments in research and development. IP encompasses patents, trademarks, copyrights, and trade secrets that enable pharmaceutical companies to safeguard their innovations, brand identities, and market positions.
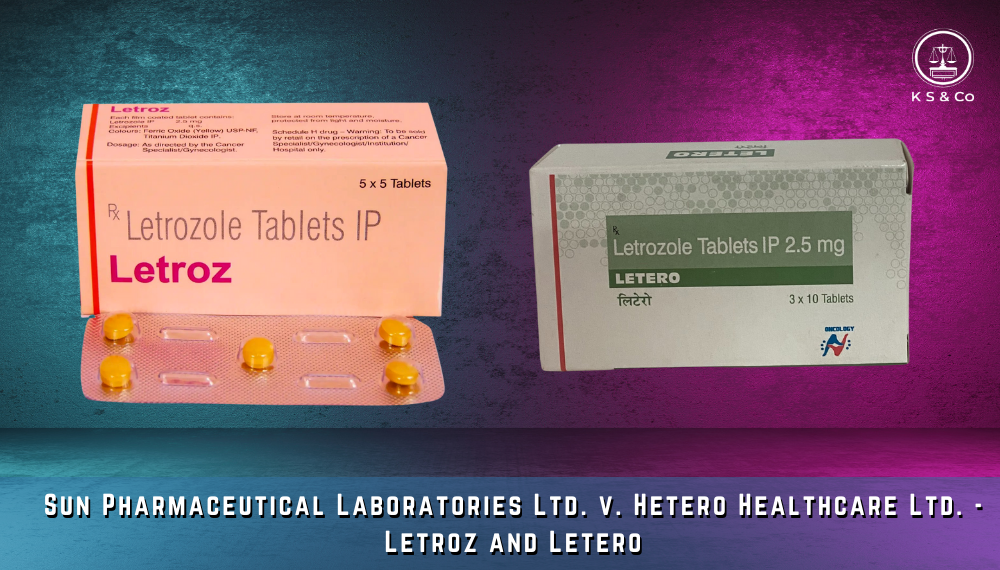
LASA issues in the pharmaceutical industry intersect with trademark laws that ensure that brand names of medicines are distinguishable. To avoid confusion and potential medication errors, pharmaceutical companies must ensure that their trademarks, product names, and packaging are distinctly different from those of competitors. This requires not only registering and defending trademarks but also carefully designing and reviewing brand elements. Effective management of these IP aspects helps prevent legal disputes, maintains clear market presence, and ensures that patients can easily identify and trust the medications they use.










Leave a Reply