The September 2025 edition of our Intellectual Property Rights Newsletter brings into focus the most significant legal developments and judicial pronouncements impacting the Indian IP and pharmaceutical landscape. This issue reviews a series of landmark cases addressing trademark disputes, clarity in patent prosecution, and enforcement of biosimilar patents in tightly regulated sectors.
Each case analysis highlights the central issues before the courts, the reasoning adopted in the judgments, and the broader commercial and legal implications for stakeholders. By examining both procedural and substantive aspects, the newsletter underscores how evolving jurisprudence continues to shape strategies for protecting and enforcing intellectual property rights in India.
Our aim is to provide readers with a concise yet comprehensive digest that not only tracks legal trends but also offers practical insights. We hope this equips businesses, innovators, and legal practitioners to anticipate challenges, make informed decisions, and strengthen their ability to safeguard innovation and brand equity in an increasingly competitive environment.
1. RSPL Health Pvt Ltd v. Sun Pharma Laboratories Limited and Anr
FAO (COMM) 65/2025 ; Hon’ble Delhi High Court ; Decided on – 12th June 2025
Background
RSPL Health Pvt Ltd, the proprietor of the well known brand “PRO-EASE” sanitary napkins, initiated a legal action against Sun Pharma Laboratories Limited regarding its use of the trademark “PRUEASE” for pharmaceutical preparations used in the treatment of chronic constipation.
 RSPL argued that “PRUEASE” was deceptively similar to its registered mark “PRO-EASE” and the similarity was sufficient to cause confusion or deception among the public. A key factor of RSPL’s case was that both the marks were registered under Class 5 of the Nice Classification, which covers a wide range of pharmaceutical and sanitary products. On that basis, RSPL contended that the similarity of marks coupled with the same class registration could mislead consumers into believing that both products had a common origin or some form of commercial association.
RSPL argued that “PRUEASE” was deceptively similar to its registered mark “PRO-EASE” and the similarity was sufficient to cause confusion or deception among the public. A key factor of RSPL’s case was that both the marks were registered under Class 5 of the Nice Classification, which covers a wide range of pharmaceutical and sanitary products. On that basis, RSPL contended that the similarity of marks coupled with the same class registration could mislead consumers into believing that both products had a common origin or some form of commercial association.
The Commercial Court (Patiala House District Court) at first instance, however, declined to grant an interim injunction in favour of RSPL, holding that the requirements for such relief had not been met. Dissatisfied with this outcome, thereafter RSPL also filed an appeal before the Hon’ble Delhi High Court.
Key Legal issues
The case raised several important questions under trademark law:
- Whether the competing marks “PRO-EASE” and “PRUEASE” were so deceptively similar that they might mislead consumer into assuming a trade connection?
- Whether the registration under Class 5 confers an exclusive right to prevent the use of any similar mark on all goods within that class, irrespective of their distinct nature?
- Whether a trademark owner can block another’s mark based on a mere possibility of expanding its business into new segments within the same class in the future?
- Whether RSPL had successfully demonstrated a prima facie case, balance of convenience, and irreparable harm – the three pillars of interim injunctions?
Court’s findings
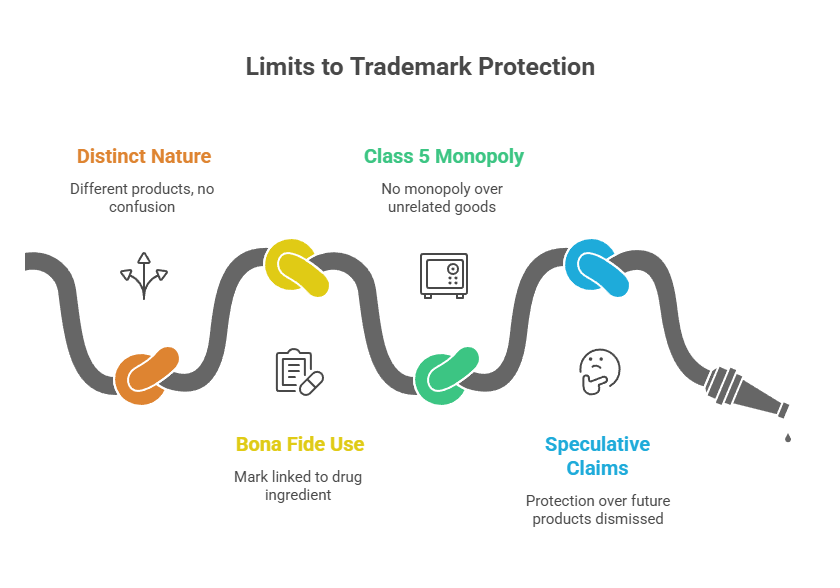
- The appeal stands rejected as the Hon’ble court found no likelihood of confusion due to distinct nature, trade channel and consumer base of sanitary napkins and constipated medicines.
- RSPL’s claims for protection over hypothetical future pharmaceutical products was dismissed as speculative.
- Sun Pharma demonstrated bona fide use of the mark “PRUEASE” since 2017, linked to its drug’s active ingredient, prucalopride.
- Mere registration within Class 5 does not grant monopoly over all unrelated good in that class.
- RSPL failed to meet the conditions to grant interim relief, including lack of a strong prima facie case and absence of irreparable harm.
Why this case matters for Companies and Organisations
- Clarifies limits of Trademark protection across product lines, emphasizing that companies cannot assume blanket protection over different goods in the same class without showing relatedness.
- Highlights the importance of bona fide uses and distinctiveness when selecting marks, especially in pharma and FMCG sectors.
- Warns against relying on speculative future business plans to claim trademark rights, encouraging more precise market focus for brand protection strategies.
Guides companies on risks of brand dilution claims where products are unrelated, and how such cases may not amount to trademark infringement.
2. Taiho Pharmaceutical Co. Ltd v. The Controller of Patents
C.A. (COMM.IPD-PAT) 6/2022 ; Hon’ble Delhi High Court ; Decided on 15th May 2025
Background
Taiho Pharmaceutical Co. Ltd filed a national phase patent application in India for a “Novel Piperidine Compound or Salt thereof”, indicated to have anti-cancer properties. The Indian Patent Office rejected the application, citing lack of inventive step (Section 2(1)(ja), Patents Act, 1970) and failure to demonstrate enhanced efficacy over prior arts D1 under Section 3(d) of the Patents Act, 1970. The Controller deemed the claimed compounds to be obvious derivatives of substances disclosed in prior art and asserted that the application did not show a significant enhancement in therapeutic efficacy.
Key legal issues
- Whether the patent application demonstrated an inventive step over prior art as required by Section 2(1)(ja) of the Patents Act, 1970?
- Whether the applicant provided evidence of enhanced efficacy over the “known substance” under section 3(d) of the Patents Act, 1970?
- Whether the controller of patents provided sufficient clarity and procedural fairness regarding the objections?
Court’s findings
- The Court found the Patent office failed to clearly and specifically identify a “known substance” from prior art D1 against which enhanced efficacy should be known.
- The Court clarified that the threshold for “enhanced efficacy” requires the Patent Office to specify precisely the known substance and explain how the claimed compound is a new form of it, thereby triggering the need for efficacy comparison. Without this clarity, applicants cannot fairly be expected to produce relevant data.
- It was insufficient to refer to a broad Markush formula or general structure similarity. A Markush claim is a type of patent claim, primarily used in chemical and biotech inventions, that covers a generic chemical structure with alternative substituents or components, allowing a single claim to protect a family of related compounds with broad potential applications. Precise identification is required so that the applicant is able to respond with applicable data.
- The court held that without clarity, the grounds for refusal under Section 2(1)(ja) and 3(d) were unsustainable. The applicant was not given a reasonable chance to respond.
- The matter was remanded for reconsideration and a fresh hearing, requiring the Patent Office to articulate specific objections and allow response.
Why this case matters for companies and organisations
- Ensures procedural clarity – The decision mandates that patent authorities must clearly specify the precise prior art and known substance when raising objections, ensuring applicants fully understand and are able to address the reasons for rejection.
- Protects applicant rights – Companies have a greater assurance that they will receive a fair opportunity to respond to substantive patentability objections, supporting more transparent, efficient patent prosecution.
- Influences R&D and Patent Strategy – By clarifying the evidentiary threshold for “enhanced efficacy” under Section 3(d), organisations can better plan data generation for patent filings and anticipate possible objections in India’s stringent IP regime.
- Reinforces best practices – The judgement encourages clear, reasoned patent examination standards, helping organisations to reduce risk of arbitrary refusals and strengthening the protection of genuine pharmaceutical innovations in India.
3. M/s Apex Laboratories Pvt Ltd vs M/s Knoll Healthcare Pvt Ltd.
C.S. (Comm. Div.) No. 355 𝘰𝘧 2020 ; Hon’ble Madras High Court ; Decided on 19th June 2025

Background
Apex Laboratories, proprietor of the registered trademark ZINCOVIT since 1988, alleged that Knoll Healthcare’s mark ZINOLVITA was deceptively similar to its own and was being marketed in packaging that closely resembled Apex’s distinctive seven-colour label. On this basis, Apex sought a permanent injunction against Knoll, claiming trademark infringement, passing off, and copyright violation.
During the pendency of the suit, Apex also filed a rectification petition challenging Knoll’s registration of the mark ZINOLVITA, arguing that it had been registered without sufficient cause and was likely to mislead consumers. In response, Knoll filed its own rectification petition, seeking to restrict Apex’s rights by preventing exclusive claims over the terms “zinc” and “vit” in ZINCOVIT.
Key legal issues
- Whether ZINOLVITA was deceptively similar to ZINCOVIT so as to cause infringement and passing off?
- Whether ZINCOVIT, though not inherently distinctive, had acquired distinctiveness through long and continuous market use?
- Whether the descriptive elements (zinc, vit) could be monopolized?
Court’s finding
- The Court held that even a non-inherent distinctive mark like ZINCOVIT could acquire strong legal protection through long and extensive use, termed “acquired distinctiveness”.
- Knoll presented no substantial evidence of widespread third party use of genericness of the trade.
- Apex could not claim exclusive rights over the use of the sperate words ‘zinc’ and ‘vit’ but could assert rights over the mark ZINCOVIT as a whole.
- The adoption and use of highly similar packaging by Knoll suggested bad faith and a likelihood of consumer confusion, especially given the identical nature of their pharmaceutical products.
- The rectification filed by Apex against Knoll’s mark was allowed. However, Knoll’s counterpetition was dismissed. A permanent injunction restrained Knoll from using ZINOLVITA.
- The Court further observed that even after Knoll changed its packaging, the mark ZINOLVITA remained deceptively similar to ZINCOVIT, amounting to passing off.
Why this case matters for companies and organisations
The judgment reinforces the principle that even inherently non-distinctive trademarks can acquire distinctiveness through long and extensive use, entitling the proprietor to protection against deceptively similar marks.
- Strengthens legal value of established brands – Companies can protect marks that have acquired market recognition through prolonged and extensive use, even if not inherently distinctive, making consistent branding a powerful shield against infringers.
- Trademark strategy clarified – Trademark rights rest in the mark as a whole. Organisations can not monopolise generic or descriptive components, but can safeguard against deceptive similarity by rivals in the same sector.
- Packaging and bad faith signals – mimicry in packaging or branding can tip cases in favour of the prior use. Careful record keeping and distinctive visual branding are essential.
- Precedent for pharmaceutical and regulated sectors – the judgement highlights the need for vigilance in trademark management and rectification tools, reducing the risk of brand dilution and consumer confusion in industries with high standards for public trust and brand equity.
4. Dabur India Limited v. Patanjali Ayurveda Limited and Anr.
C.S. (Comm. Div.) No. 1195 𝘰𝘧 2024; Hon’ble Delhi High Court ; Decided on 3rd July, 2025
Background
Dabur India Limited (“Dabur”) filed suit against Patanjali Ayurved Limited (“Patanjali”), alleging that Patanjali’s television commercials and print advertisements made false and misleading statements, disparaging Dabur’s “Chyawanprash” product. Dabur contended the ads created a misleading impression alleging that only Patanjali had authentic Ayurvedic knowledge, and other Chyawanprash, were ‘ordinary’ or not prepared in accordance with tradition. Patanjali argued its ads constituted puffery and permissible comparative advertising and was not in any way disparaging.
Key legal issues
- Whether Patanjali’s advertisements constituted unlawful disparaging versus permissible comparative advertisement or puffery?
- Whether it is permissible under the law to claim exclusive Ayurvedic knowledge in advertising regulated drugs?
- The effect of direct or indirect reference to a competitor’s product in advertising, especially regarding regulated health products.
Court’s findings
- The court held that while comparative advertising is permissible and protected under the right to commercial speech (Article 19(1)(a), Constitution of India), this does not extend to misleading claims that disparage competitor’s products or mislead consumers regarding the efficacy or safety of the competitor’s products.
- The threshold of advertisement is higher when advertisements relate to health or regulated products, with courts required to examine if claims may affect consumer decisions detrimentally.
- The court found the impugned ads unmistakably identified and disparaged Dabur’s product, both specially and generically (‘ordinary Chyawanprash”) and misrepresented the basis for ‘superiority’, thus amounting to disparagement and unfair trade practice.
- The court restrained Patanjali from publishing or airing the disputed advertisements, ordering deletion of statements disparaging Dabur’s products, and disallowed future similar representations.
Why this case matters for companies and organisations
- Clarifies standards on comparative advertising – brands may highlight their strengths, but cannot disparage competitors or make misleading claims, especially about regulated or health related products.
- Raises bar for advertising health products – courts will scrutinize claims and representations in pharma and wellness advertising more strictly, imposing higher standards of truthfulness and fairness.
- Supports brand reputation defence – established market leaders have recourse when misleading or disparaging ads target their products.
5. R. Squibb and Sons LLC & Ors vs Zydus Lifesciences Limited.
C.S. (Comm. Div.) No. 376 𝘰𝘧 2024; (Hon’ble Delhi High Court); Decided on 18th July, 2025
Background
E.R. Squibb and Sons LLC holds Indian Patent No. IN 340060 covering the monoclonal antibody Nivolumab, marketed globally as Opdivo, used for cancer treatment by targeting the PD-1 immune checkpoint protein. Zydus Lifesciences developed a biosimilar named ZRC-3276, referencing Nivolumab in clinical trials.
Squibb filed a quia timet action to restrain Zydus from commercially launching the biosimilar before the patent expiry date 2nd May, 2026, alleging infringement despite Zydus’s claim of protection under India’s Bolar exemption (Section 107A of the Patents Act, 1970).
Key Legal Issues
- Whether the biosimilar developed by Zydus infringed Squibb’s patent, was based on claim scope and structural similarity including Complementarity Determining Regions (CDRs)?
- What is the applicability and scope of the Bolar Exemption regarding research, trials, and stockpiling before patent expiry?
- What is the evidentiary threshold for quia timet injunctions in anticipation of infringement?
- What is the role and weight of Opposition Board Recommendations (OBR) in patent validity assessments?
Court’s finding
- The Court held the patent valid and subsisting, recognizing Squibb’s patent as a product patent, distinct from prior art cited by Zydus.
- It found a strong prima facie case of infringement due to the identical amino acid sequences (CDRs) shared by the biosimilar and patented Nivolumab.
- The Court rejected Zydus’s reliance on the non-binding OBR and held that biosimilar under regulatory guidelines does not negate patent infringement if the claims cover the structural features of biosimilar.
- Bolar exemption (Section 107A of the Patents Act, 1970 provides for Bolar exemption. This exemption allows manufacturers to make, use, and even export patented inventions strictly for regulatory purposes, to ensure post-patent market entry without any delay) was acknowledged for clinical trials but did not extend to commercial launch preparations or stockpiling aimed at market entry before patent expiry.
- The court decided it was more important to protect Squibb’s patent, since Zydus selling the drug without permission could cause serious and unfair harm to Squibb.
Why this case matters for companies and organizations
- Injunctions for biosimilars – Courts in India may stop biosimilars at an early stage, even before launch, if regulatory filings show the product is too close to a patented drug.
- Limits of the Bolar exemption – Research and trials are allowed, but stock piling or preparing for sales without consent can still count as patent infringement.
- Evidence needed – Courts may assume infringement if a biosimilar is very similar to the patented drug, without needing a full technical breakdown.
- Public health impact – The ruling makes it harder for generics and biosimilars to enter the market early, which protects innovation but may delay cheaper medicines
- Opposition Board Recommendations are not final (In Patent law, an Opposition Board Recommendation is a report provided by a panel of experts (the Opposition Board) to the Controller of Patents, following their examination of a patent opposition) – Courts have the last word, so strong legal challenges remain important even after the opposition stage.
6. Mankind Pharma Ltd. v. Ram Kumar M/s Dr. Kumars Pharmaceuticals
C.O. (COMM.IPD-TM) 566/2022 ; Delhi High Court ; Decided on 22nd August, 2025
Background
Mankind Pharma Ltd., a prominent pharmaceutical company and proprietor of the “MANKIND” mark and its well-known “KIND” family of trademarks, sought rectification and removal of the mark “UNKIND” (registered in Class 35 by the respondent) from the Trademarks Register. The petition was based on long-standing use, extensive portfolio (over 300 registrations with the “KIND” element), and the assertion that the impugned mark was never commercially used by the respondent. The mark “UNKIND” had been registered since 2008, but with no evidence of bona fide use or market presence.
Key Legal Issues
- Whether the registration of “UNKIND” was valid despite its non-use and similarity to Mankind’s established “KIND” family of marks?
- Whether continued registration of “UNKIND” risked confusion and dilution of the distinctive character and goodwill built around Mankind’s marks?
- Application of the “family of marks” doctrine. Does prior, extensive use and public association with the “KIND” suffix entitle Mankind to higher trademark protection?
- Scope of rectification under Sections 47, 57 (removal for non-use and wrongful entry) and Section 11 (likelihood of confusion) of the Trade Marks Act, 1999.
Court’s Finding
- The Court found that Mankind Pharma was a prior user and registered proprietor of a well-known family of “KIND” marks, enjoying extensive goodwill and public association through decades of continuous use.
- The respondent failed to appear and offer any evidence of bona fide use; the pleadings stood uncontested and were deemed admitted.
- The Court reaffirmed that the word “KIND”, though inherently unrelated to pharmaceuticals, had, through extensive and consistent use, become uniquely and strongly associated with Mankind’s goods.
- The “UNKIND” mark, by mimicking the “KIND” family, was likely to confuse the public and trade upon Mankind’s reputation. Registration was held to be lacking in distinctiveness as well as bona fide use, and liable for cancellation under Sections 47 and 57 of the Trademark Act.
- The Court did not grant a blanket monopoly over “KIND” but made a specific finding of deceptive similarity and bad faith for “UNKIND”, consistent with the refined “family of marks” doctrine.
Key Concepts Explained
Doctrine of Family of Marks
When a business owns a collection of trademarks sharing a common element (like “KIND”), courts provide heightened protection against competitors who attempt to adopt deceptively similar marks. Even if a competitor is not copying a specific registered mark, the law recognizes the risk of confusion across the entire brand “family.” In this case, Mankind’s consistent use of “KIND” (e.g., MANKIND, PREGAKIND, HEALTHKIND) meant the public strongly associated the suffix with Mankind’s products, making “UNKIND” deceptively similar.
Series Trademark
A series trademark allows companies to register related marks that share a distinctive element while differing only in minor aspects. For example, Mankind’s registrations such as “CARDIOKIND” and “HEALTHKIND” protect the “KIND” element across multiple product lines. This legal tool strengthens enforcement against imitators and prevents fragmentation of brand identity.
Warehousing of marks
The Court highlighted the risk of warehousing, where a party registers marks without genuine commercial use, often to block others from registering similar marks. The “UNKIND” registration fell into this category, as the respondent had no evidence of market presence or bona fide use since 2008. Under Sections 47 and 57 of the Trademarks Act, such marks are vulnerable to cancellation, reinforcing that trademark rights depend on actual use, not just registration.
Why this case matters for Companies and Organisation
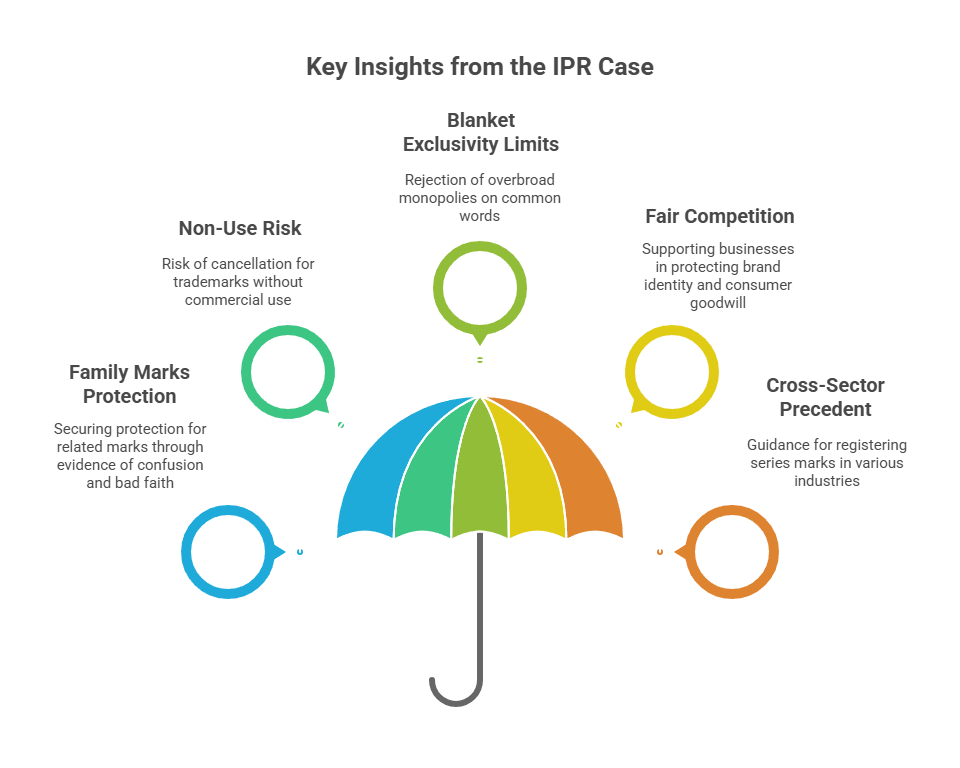
- Case-by-case protection for “family marks”: Businesses with a portfolio of related marks can secure heightened legal protection when they show long-term, exclusive use and market recognition, but each enforcement case requires evidence of risk of actual confusion among consumers and competitor’s bad faith.
- Non-use and warehouse registrations at risk: The case underscores that “warehousing” trademarks for defensive purposes without bona fide commercial use can lead to cancellation. This is vital for companies relying on broad portfolios without having actual use.
- Guardrails on blanket exclusivity: The ruling rejects overbroad monopolies on common words (like “KIND”), instead supporting evidence-based, context-specific enforcement resulting in strategic brand portfolio management.
- Reinforces fair and defensible competition: The judgment supports businesses in protecting the true source identity and consumer goodwill they have built, while also ensuring robust competition and limiting abuse of common language in trademarks.
- Cross-sector precedent: The approach taken by the Court of balancing the family of marks doctrine with anti-monopoly safeguards, serves as essential guidance for organizations in pharma and other brand-driven industries to consider registering for “series” marks.
7. Boehringer Ingelheim International GmbH & Anr. v. Femilab Healthcare & Anr.
OMP No.1085 of 2024 in COMS No.24 of 2024 ; High Court of Himachal Pradesh ; Decided on – 29th August, 2025
Background
Boehringer Ingelheim International GmbH, the patentee of Indian Patent No. 268846 covering the diabetes drug Empagliflozin, filed a patent infringement suit seeking interim relief against Femilab Healthcare and others, alleging unauthorized manufacture, sale, and distribution of infringing Empagliflozin products. The Court initially granted an interim injunction in December 2024 restraining the defendants from infringing the patent during its subsistence. However, as the patent expired on 11 March 2025, the continued applicability of the injunction was questioned.
Key Legal Issues
- Whether an interim injunction granted for patent infringement can continue to operate after the patent’s expiry?
- The legal consequences for products manufactured or imported during the patent term but marketed after patent expiry.
- The balancing of patent holders’ interests with public policy allowing free market entry post-patent.
Court’s Findings
- The Court held that interim injunctions granted to protect patent rights serve to prevent infringement during the patent’s life and do not survive beyond patent expiry.
- It acknowledged that while infringement occurring within the patent term remains actionable and can justify restraint on products manufactured during that period, the injunction as a protective order cannot continue post-expiry.
- Citing the Supreme Court’s ruling in Novartis AG & Anr. v. Natco Pharma Limited (Special Leave Petition (Civil) No.16237 of 2024), the Court emphasized that once a patent expires, it ceases to confer monopoly rights, and the market must be open to competitors.
- The Court declined the plaintiff’s plea to extend the injunction beyond expiry, observing that no legal provision supports restricting post-patent entry into the market.
- The injunction was accordingly vacated, with explicit directions that infringing products produced during the patent’s life remain subject to restraint from being marketed without authorization only during validity of the patent.
Why this Case matters for Companies and Organisation
- Defines the time limit of patent injunctions: Interim injunctions apply only while the patent is valid. Once a patent expires, competitors are free to enter the market.
- Encourages fair competition after expiry: After the patent term ends, companies may legally manufacture and sell generic or equivalent products, promoting competition and accessibility.
- Holds infringers accountable before expiry: Any unauthorized use of the patent before its expiry can still be challenged, preventing attempts to bypass patent rights.
- Guides enforcement strategies: Patent holders should plan legal action carefully and act before expiry, as injunctions cannot extend beyond the patent term.
- Assists business and regulatory planning: Especially in pharmaceuticals, companies can time product launches and marketing with patent expiries to reduce legal risks.






